twinSys uncemented
MATHYS
twinSys uncemented
The basic design of the twinSys stem system is based on the straight-stem prosthesis concept by Prof. Maurice E. Müller. The twinSys system has been distributed since 2003 with the aim of enabling surgeons to perform cemented and uncemented implan-tation with a single instrumentation.
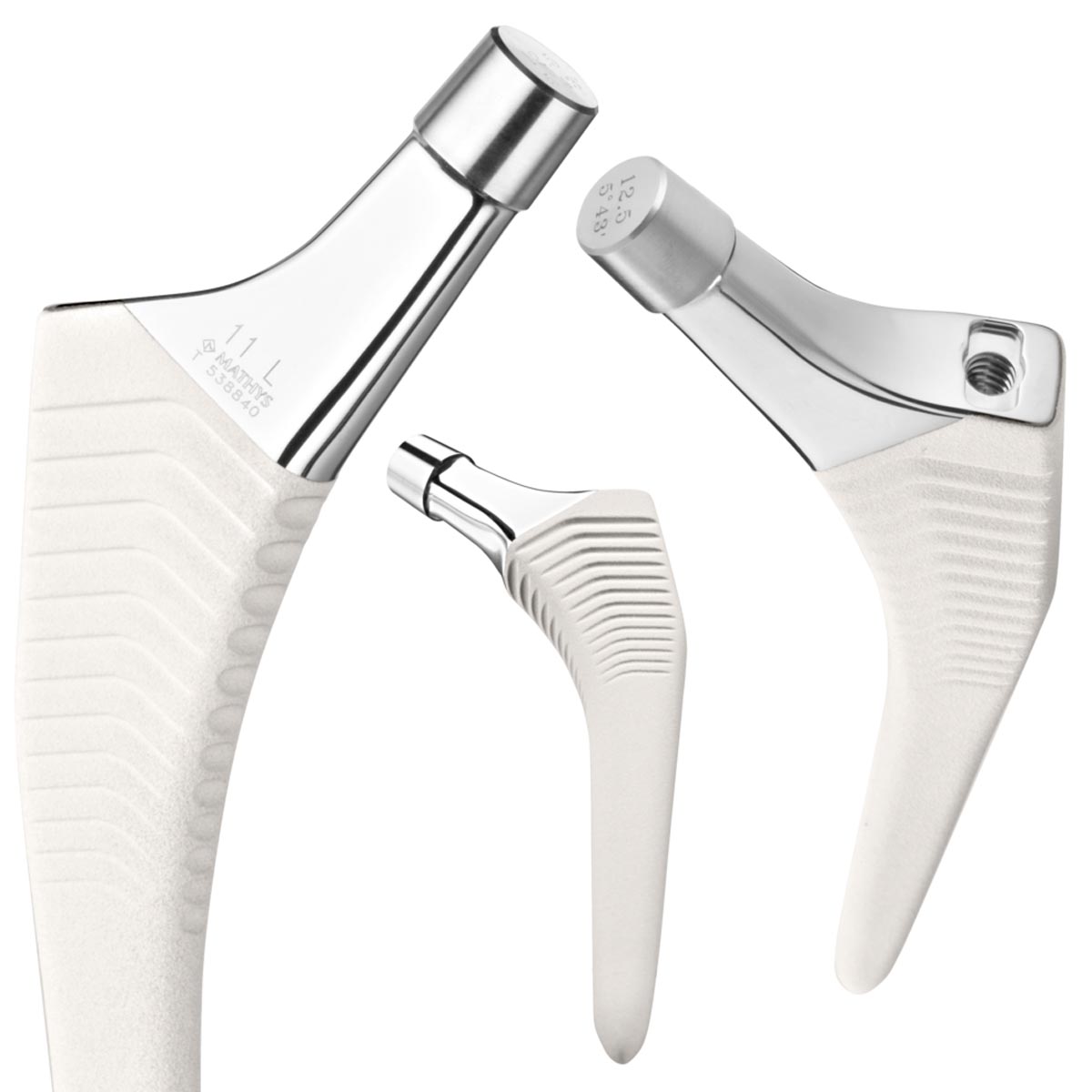
The uncemented twinSys stem is a straight monoblock prosthesis available in XS, Standard, Lateral and Long versions. It has a triply conical stem geometry and a pronounced proximal rib structure to counteract forces acting axially on the stem. The stem design strives to achieve primary pressfit anchoring in the proximal cancellous bone. Data from a study conducted specifically to this end show that the twinSys stem design reduces the risk of postoperative subsidence and provides secondary stability.
The selected cross-sectional geometry increases the stability of the implant against rotational forces acting on it.1 In order to counteract premature distal stem locking and reduce the risk of femoral pain, the stem is provided with a tapered distal prosthesis tip.
Using the plasma spray process, a hydroxyapatite coating is applied to the corundum-blasted surface in order to support osseointegration of the implant into the bone.2
The optimys stem, in combination with a Mathys ceramic head and the RM Pressfit vitamys cup, is known as the bonepreservation system.
Enquire
For further information, questions regarding this product, or to discuss alternative solutions, please get in touch with your local Sales Specialist or our Head Office using the form or the contact details at the bottom of the page.
Stay in touch
NEWSLETTER
Sign up to receive email updates on new product announcements, insights on surgical techniques from surgeons, specialists, and sales representatives and industry trends, such as changes in regulations and new research findings.